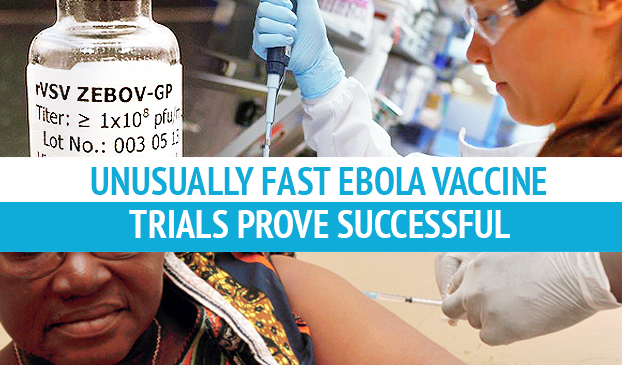
An Ebola vaccine was found to be 100 percent successful, but additional large-scale trials need to take place before its effectiveness is confirmed.
The epidemic in West Africa may finally come to an end after a new drug proved 100 percent successful in recent trials. Research that would have normally taken a decade was completed in just 12 months, which means the speed of the drug’s development is unprecedented.
The rVSV-ZEBOV vaccination came from a collaboration between various scientists, doctors, donors and drug companies. The World Health Organization took on the risk of sponsoring the trials, which marks another unusual element in this promising but out-of-the-ordinary drug advancement.
The Trials

Tests to prove the Ebola vaccine effective were completed in just 12 months in an extremely fast round of drug trials.
Via: Cidrap
The trials were conducted in Guinea and included 4,000 people. The outbreaks have recently shifted and become regional in nature, which created a perfect structure for testing the new vaccine. Researchers employed a “ring” trial design, where all individuals in each of the contaminated clusters were given the drug.
In the villages where ebola was present, researchers vaccinated the family, friends and neighbors who came into contact with the sick person, except for children, adolescents and pregnant women to ensure their safety. About 50 percent of villagers received the vaccination.
Some clusters were given the vaccination before being diagnosed with ebola. 2,014 people were vaccinated immediately and none developed the sickness. They were tested after 10 days had passed and their bodies had been able to develop an immunity. Other clusters were tested with a delayed vaccination method after infection was introduced. Out of 2,380 people, only 16 cases were reported.
“This trial dared to use a highly innovative and pragmatic design, which allowed the team in Guinea to assess this vaccine in the middle of an epidemic,” said Jeremy Farrar, director of the Wellcome Trust and one of the world’s leading experts on infectious disease. “It is a remarkable result which shows the power of equitable international partnerships and flexibility.”
The Supporters

The U.S., London, Norway, Canada, France, and Guinea collaborated to create the vaccine.
Via: Herzen
The drug is truly a result of widespread international cooperation. The vaccine was originally developed by the Public Health Agency of Canada. Now, the vaccine is being tested and produced by Merck & Company, an American pharmaceutical company.
London’s biomedical research charity Wellcome Trust, the governments of Norway and Canada, Médecins sans Frontières from France, and the London School of Hygiene and Tropical Medicine came together to fund the vaccine. The majority of the staff who conducted the trials were from Guinea, which was 90 percent responsible for carrying out its first round of clinical research.
“This partnership also shows that such critical work is possible in the midst of a terrible epidemic. It should change how the world responds to such emerging infectious disease threats,” Farrar said.
The Trials Continue

The trials were conducted in Guinea by a primarily domestic medical staff.
Via: Humanosphere
The trials garnered promising results that rVSV-ZEBOV could be the vaccine that will stop future outbreaks before they start. However, trials will have to continue on a larger scale before anything is confirmed. From now on, all individuals who have come into contact with ebola-sickened persons will be offered the vaccine. It is also been approved for use in children and adolescents.
Other potential vaccines, one from GlaxoSmithKline and the other from Johnson & Johnson, may or may not be tested.
The trial for the Merck vaccine was a race against time, but other infected countries are encouraged to immediately replicate ring vaccinations to stop transmission and protect front-line workers.
“This new vaccine, if the results hold up, may be the silver bullet against Ebola, helping to bring the current outbreak to zero and to control future outbreaks of this kind,” said Børge Brende, the foreign minister of Norway.
CitizensReport
Latest posts by CitizensReport (see all)
- 4 Natural Alternatives To Energy Drinks That Will Make You Feel Healthier - February 19, 2018
- NBC Nightly News Publishes Expose On IVC Filters - February 18, 2018
- Good Sugar, Bad Sugar: Why You Should Eat More Fruit - February 16, 2018


Join the discussion